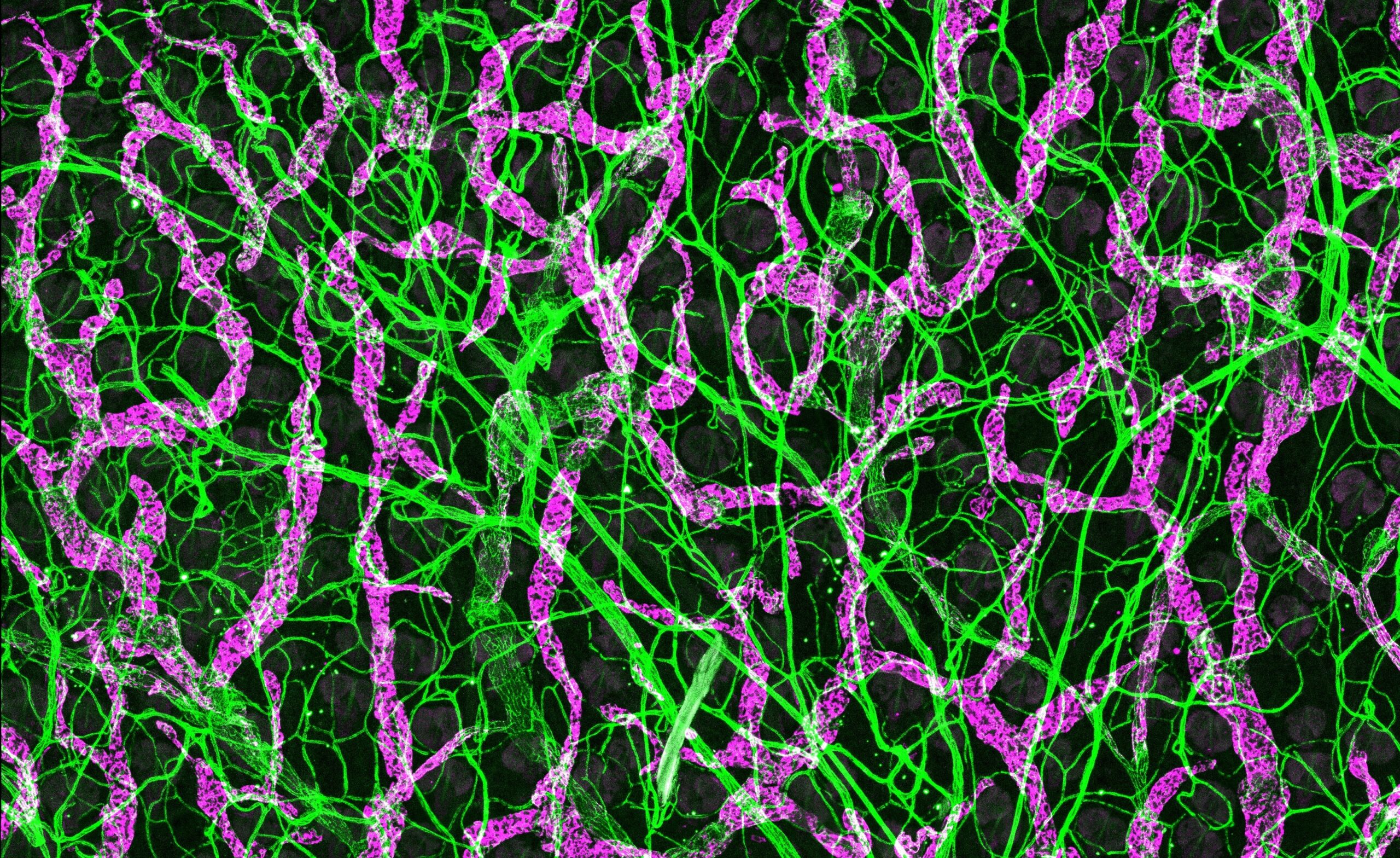
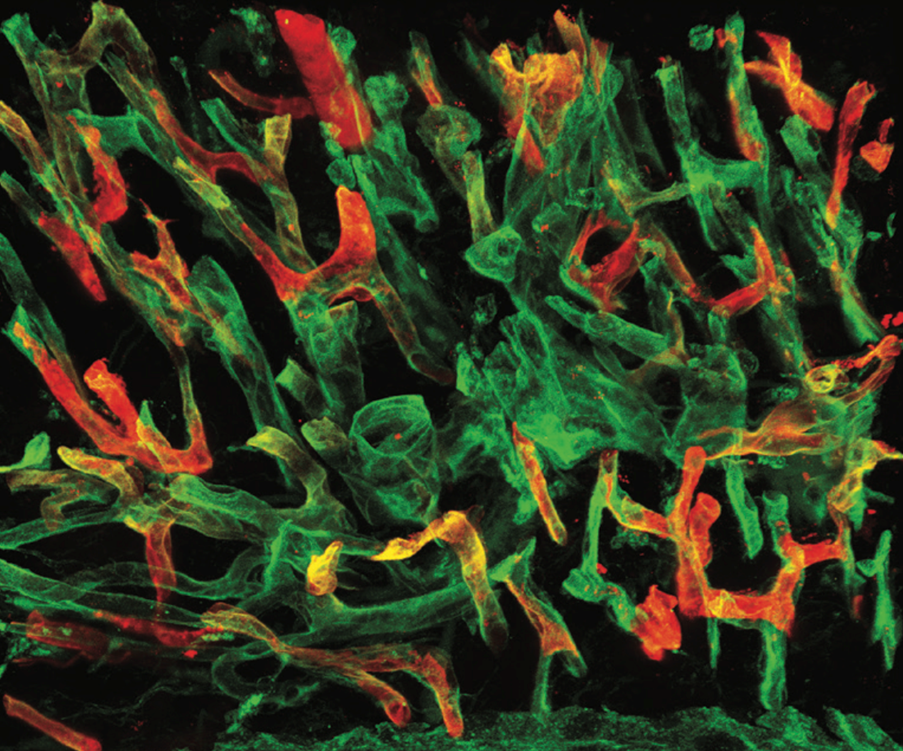
We study the fundamental mechanisms that regulate the formation and maintenance of the blood and lymphatic vascular systems in the body, and explore how these mechanisms are impaired in severe inflammation causing fluid leakage into tissues in various difficult-to-treat diseases.
Endothelial cells are key components of vascular systems, forming the inner lining of both blood and lymphatic vessels. The endothelial cells in our bodies form a semipermeable barrier estimated to cover an area comparable to that of six tennis courts. Malfunction or impaired integrity of the endothelial cell layer causes fluid accumulation in tissues. Fluid leakage from the blood circulation can impair blood perfusion and cause edema, which may lead to organ failure, for example in sepsis. However, stopping vessel leakage or improving lymphatic drainage remains a challenge in many diseases.
Comprehensive understanding of what causes vessel leakiness and how leaky vessels can be repaired, is incomplete. By studying the fundamental principles of how blood and lymphatic vessels are formed and how their stability is maintained, we aim to understand the mechanisms that cause vessel leakiness or malfunction in various human diseases. New understanding of mechanisms regulating vascular stability and proper functioning of lymphatic vessels facilitates translational research to improve vascular stability and function in various diseases.
Current Group members
Anne Pink, postdoc
Kumar Ponna, postdoc
Martina Trevisani, postdoc
Keerthana Ganesh, PhD student
Shraman Bohra, PhD student
Pihla Kaipainen, PhD student
Klaudia Lewna, PhD student
Qina Liu, PhD student
Emad Arbash, undergrad student
Saana Silvennoinen, undergrad student
Maija Arness, undergrad student
Mannion AJ., Zhao H., Zhang Y., von Wright Y., Bergman O., Björck HM., Saharinen P., Holmgren L. The junctional mechanosensor AmotL2 regulates YAP promotor accessibility. BioRxiv. 2013. doi: https://doi.org/10.1101/2023.01.13.523596
Tuohinto K, DiMaio TA, Kiss EA, Laakkonen P, Saharinen P, Karnezis T, Lagunoff M and Ojala PM. KSHV infection of endothelial precursor cells with lymphatic characteristics as a novel model for translational Kaposi’s sarcoma studies. PLoS Pathog. 2023 Jan 23;19(1):e1010753. doi: 10.1371/journal.ppat.1010753
Korhonen EA., Murtomäki A., Jha SK., Anisimov A., Pink A., Zhang Y., Stritt S., Liaqat I., Stanczuk L., Alderfer L., Sun Z., Kapiainen E., Singh A., Sultan I., Lantta A., Leppänen VM., Eklund L., He Y., Augustin HG., Vaahtomeri K., Saharinen P., Mäkinen T., Alitalo K. Lymphangiogenesis requires Ang2/Tie/PI3K signaling for VEGFR3 cell-surface expression. J Clin Invest. 2022 Aug 1;132(15):e155478. doi: 10.1172/JCI155478
Leppänen VM., Brouillard P., Korhonen EA., Sipilä T., Jha SK., Revencu N., Labarque V., Fastré E., Schlögel M., Ravoet M., Singer A., Luzzatto C., Angelone D., Crichiutti G., D’Elia A., Kuurne J., Elamaa H., Koh GY., Saharinen P., Vikkula M., Alitalo K. Characterization of ANGPT2 mutations associated with primary lymphedema. Sci Transl Med. 2020 Sep 9;12(560):eaax8013. doi: 10.1126/scitranslmed.aax8013
Hakanpaa L., Kiss EA., Jacquemet G., Miinalainen .I, Lerche M., Guzmán C., Mervaala E., Eklund L., Ivaska J., Saharinen P. Targeting β1-integrin inhibits vascular leakage in endotoxemia. Proc Natl Acad Sci USA. 2018 Jul 10;115(28):E6467-E6476. doi: 10.1073/pnas.1722317115
Pekkonen P., Alve S., Balistreri G., Gramolelli S., Tatti-Bugaeva O., Paatero I., Niiranen O., Tuohinto K., Perälä N., Taiwo A., Zinovkina N., Repo P., Icay K., Ivaska J., Saharinen P., Hautaniemi S., Lehti K., Ojala PM. Lymphatic endothelium stimulates melanoma metastasis and invasion via MMP14-dependent Notch3 and β1-integrin activation. Elife. 2018 May 1:7:e32490. doi: 10.7554/eLife.32490
Korhonen EA., Lampinen A., Giri H., Anisimov A., Kim M., Allen B., Fang S., D’Amico G., Sipila T., Lohela M., Strandin T., Vaheri A., Ylä-Herttuala S., Koh GY., McDonald DM., Alitalo K., Saharinen P. Tie1 controls angiopoietin function in vascular remodeling and inflammation. J Clin Invest. 2016 Sep 1;126(9):3495-510. doi: 10.1172/JCI84923
Hakanpaa L., Sipila T., Leppanen VM., Gautam P., Nurmi H., Jacquemet G., Eklund L., Ivaska J., Alitalo K., Saharinen P. Endothelial destabilization by angiopoietin-2 via integrin β1 activation. Nat Commun. 2015 Jan 30:6:5962. doi: 10.1038/ncomms6962