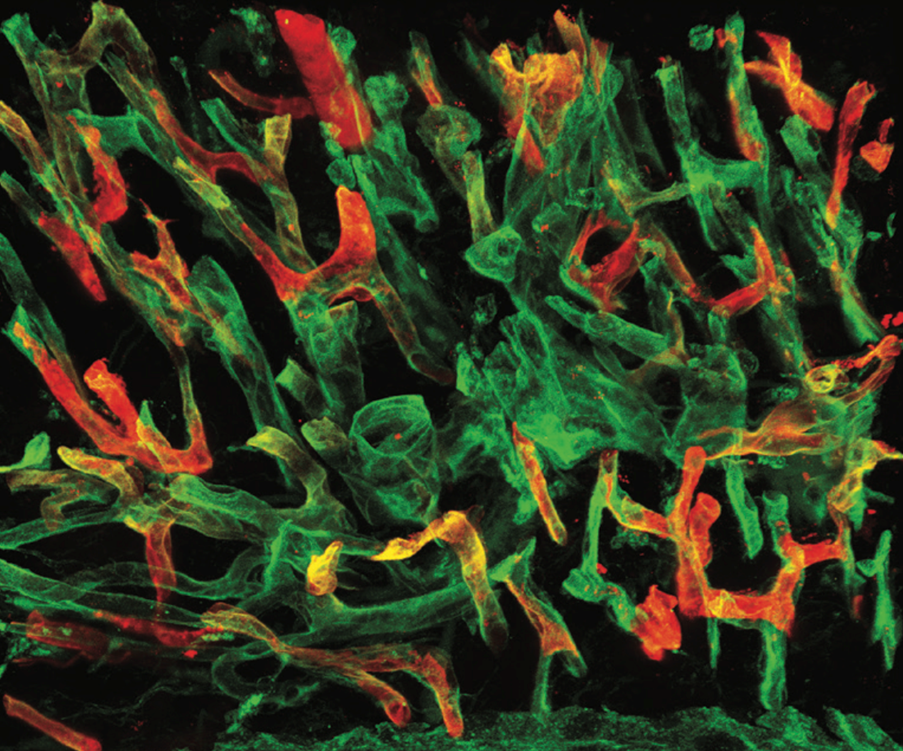
We study blood and lymphatic vessels across organs, with a particular focus on how the vessels and the endothelial cells are affected in metabolic diseases such as obesity and diabetes.
Vessels are highly specialized: depending on their location, they adopt distinct structures and functions to meet the needs of each tissue. Understanding how these organ-specific (or organotypic) properties are established, maintained, and altered in disease is central to our work.
One of the current focus areas of our work is how the structure and function of adipose tissue vasculature adapt -or fail to adapt- during metabolic stress. We examine both molecular and morphological changes in vessels of the adipose tissues, including processes such as capillary rarefaction and alterations in vessel organization, endothelial cell size and molecular properties. To study these, we use mouse models of metabolic disease alongside patient-derived samples, allowing us to connect mechanistic insights with patient-relevant biology. Our work integrates advanced imaging, molecular profiling, single-cell RNAsequencing, proteomics, and functional assays, including live imaging approaches to study blood flow dynamics in real time.
Another focus of our research is to understand whether endothelial cells from different tissues have intrinsic properties that influence parenchymal cell differentiation. We examine how these properties change in metabolic disease and how vascular pathology impacts tissue function, while also considering how local microenvironments might shape vascular behavior. To address these questions, we combine multi-omics approaches with experimental models, including adipose organoid systems to study vascular–tissue interactions in controlled settings. We work closely with clinical collaborators to ensure that our research remains connected to patient-relevant questions and supports future translational efforts.
In parallel, we investigate how metabolic stress affects vasculature across multiple organs at the whole-body level. Our work shows that obesity induces organ-specific vascular remodeling, with distinct structural and cellular changes emerging over time. These adaptations do not always follow uniform patterns across tissues, highlighting differences in how vascular networks respond to metabolic challenges. Together, these findings emphasize the importance of considering organotypic vascular responses when studying disease progression.
Overall, our goal is to define how vascular dysfunction contributes to metabolic disease progression and its complications. By identifying the principles that govern organotypic vascular responses, we aim to support the development of strategies that preserve vascular health and improve outcomes in metabolic disorders.
Current Group members
Emmi Pakarinen, Postdoctoral fellow
Nirajan Neupane, Postdoctoral fellow
Satu Paavonsalo, Doctoral student
Yelin Subashi, Doctoral student
Madeleine Lackman, Doctoral student
Marco Ahvenlammi, Technical assistant
Hanna Ruddock, Master’s student
High-fat diet feeding induces organ specific vascular remodeling with distinct temporal dynamics in male mice. Pakarinen E, Paavonsalo S, Lackman MH, Subashi Y, Ruddock HM, Karaman S. In Press. Communications Biology.
Anisimov, A., M.H. Lackman, H.G. Augustin, E. Mervaala, K. Alitalo, and S. Karaman. 2026. Deletion of the angiopoietin receptor Tie2 enhances proliferation and sprouting of cardiac endothelial cells. Angiogenesis. 29:13. doi:10.1007/s10456-025-10028-2.
Karaman, S., S. Lehti, C. Zhang, M.-R. Taskinen, R. Käkelä, A. Mardinoglu, H. Brorson, K. Alitalo, and R. Kivelä. 2024. Multi-omics characterization of lymphedema-induced adipose tissue resulting from breast cancer-related surgery. FASEB J. 38:e70097. doi:10.1096/fj.202400498RR.
Lackman, M.H., Y. Subashi, and S. Karaman. 2022. A closer look at adipose tissue lymphatics and their markers. Curr Opin Hematol. 29:144–150. doi:10.1097/MOH.0000000000000712.
Karaman, S., S. Paavonsalo, K. Heinolainen, M.H. Lackman, A. Ranta, K.A. Hemanthakumar, Y. Kubota, and K. Alitalo. 2022. Interplay of vascular endothelial growth factor receptors in organ-specific vessel maintenance. J Exp Med. 219:e20210565. doi:10.1084/jem.20210565.
Paavonsalo, S., S. Hariharan, M.H. Lackman, and S. Karaman. 2020. Capillary Rarefaction in Obesity and Metabolic Diseases-Organ-Specificity and Possible Mechanisms. Cells. 9:2683. doi:10.3390/cells9122683.
Antila, S., S. Karaman, H. Nurmi, M. Airavaara, M.H. Voutilainen, T. Mathivet, D. Chilov, Z. Li, T. Koppinen, J.-H. Park, S. Fang, A. Aspelund, M. Saarma, A. Eichmann, J.-L. Thomas, and K. Alitalo. 2017. Development and plasticity of meningeal lymphatic vessels. J Exp Med. 214:3645–3667. doi:10.1084/jem.20170391.
Heinolainen, K., S. Karaman, G. D’Amico, T. Tammela, R. Sormunen, L. Eklund, K. Alitalo, and G. Zarkada. 2017. VEGFR3 Modulates Vascular Permeability by Controlling VEGF/VEGFR2 Signaling. Circ Res. 120:1414–1425. doi:10.1161/CIRCRESAHA.116.310477.
Karaman, S., M. Hollmén, S.-Y. Yoon, H.F. Alkan, K. Alitalo, C. Wolfrum, and M. Detmar. 2016. Transgenic overexpression of VEGF-C induces weight gain and insulin resistance in mice. Sci Rep. 6:31566. doi:10.1038/srep31566.
Karaman, S., M. Hollmén, M.R. Robciuc, A. Alitalo, H. Nurmi, B. Morf, D. Buschle, H.F. Alkan, A.M. Ochsenbein, K. Alitalo, C. Wolfrum, and M. Detmar. 2015. Blockade of VEGF-C and VEGF-D modulates adipose tissue inflammation and improves metabolic parameters under high-fat diet. Mol Metab. 4:93–105. doi:10.1016/j.molmet.2014.11.006.