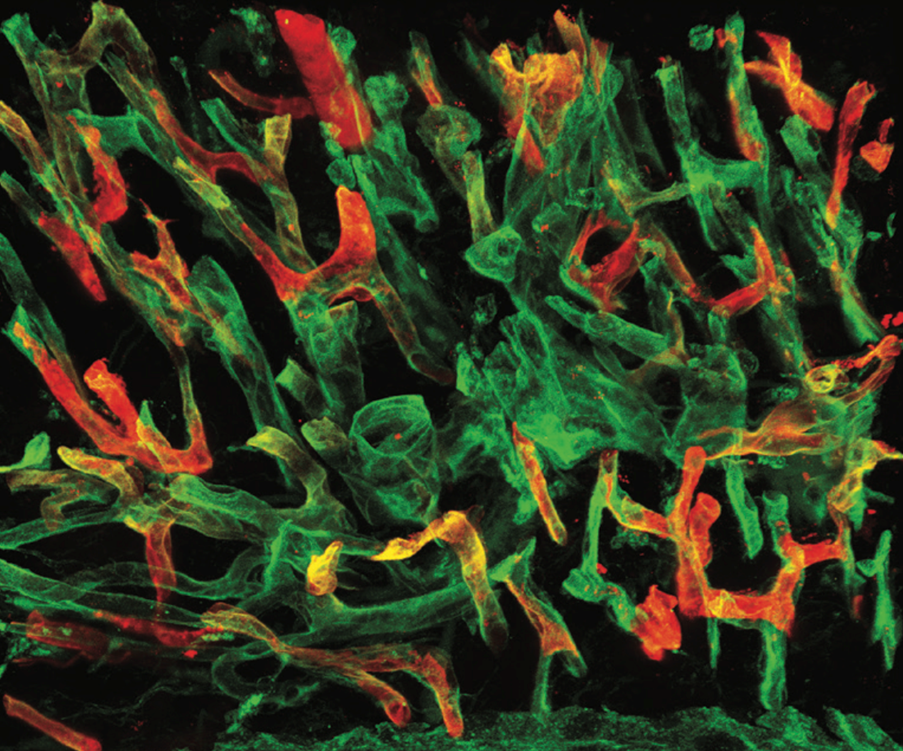
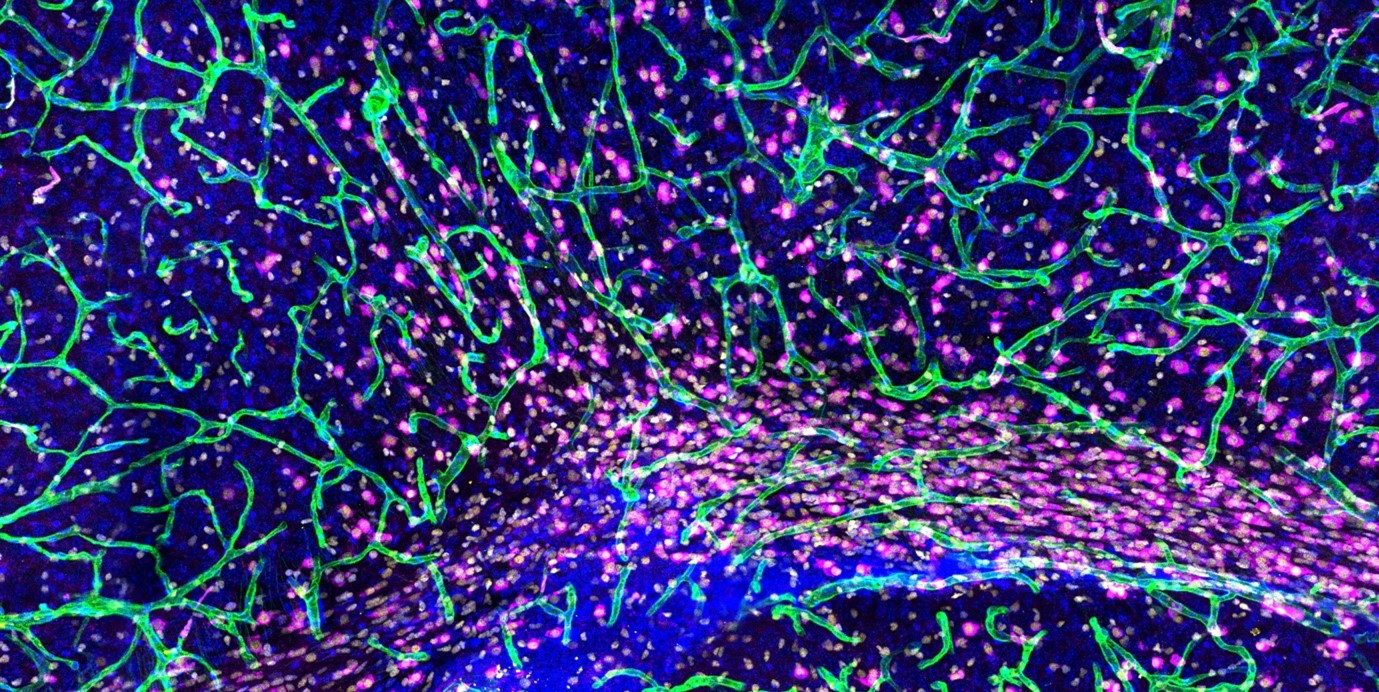
We study the mechanisms governing the development, maintenance, and regeneration of the lymphatic vasculature, and how its function and dysfunction contribute to organ-specific physiology and disease.
The lymphatic vasculature is increasingly recognized as a multifaceted regulator of tissue homeostasis and regeneration. Traditionally, its primary function has been to drain fluid, macromolecules, and immune cells from peripheral tissues into the systemic circulation via lymph nodes. Dysfunction of lymphatic vessels can lead to the accumulation of protein-rich fluid in tissues (lymphedema) and impaired immune responses. More recently, additional roles of the lymphatic system have emerged, including active modulation of adaptive immunity by lymphatic endothelial cells (LECs) and the secretion of paracrine (lymphangiocrine) factors that regulate organ growth and regeneration. These diverse functions are implicated in diseases such as autoimmune disease and atherosclerosis, underscoring the need for a better understanding of the underlying mechanisms.
Our research aims to uncover the cellular and molecular mechanisms that regulate lymphatic vessel growth, specialization, and function across tissues, and to understand how their dysfunction contributes to disease onset and progression. This knowledge provides a basis for developing novel therapeutic strategies targeting lymphatic vasculature.
A major focus of our work is to uncover tissue-specific functions of LECs in health and disease. Single-cell transcriptomics has revealed substantial heterogeneity among LECs, including the identification of a specialized PTX3+ immune-interacting subpopulation in dermal lymphatic capillaries (Petkova et al, JEM 2023). These cells contribute to lymphatic malformations by promoting immune cell recruitment and pathological vessel growth through paracrine signaling. Together, these findings highlight a dynamic crosstalk between lymphatic and immune cells and suggest broader roles for LEC-derived signals in the tissue-specific regulation of development and disease, which we continue to investigate.
In parallel, we investigate mechanisms of lymphatic vessel resilience in vivo. Using advanced intravital imaging approaches, we study LEC behavior and dynamics in living tissues at single-cell resolution. Our work has shown that cytoskeletal regulation of LEC shape is critical for maintaining vessel integrity under physiological conditions (Schoofs et al, Nature 2025). Ongoing studies aim to further define how lymphatic vessels balance structural stability with controlled permeability to ensure efficient fluid and immune cell transport.
Current Group members
Abiodun Ayo, postdoc
Gauri Arolkar, PhD student
Ingvar Ferby, researcher
Vishal Mohanakrishnan, postdoc
Dmitry Molotkov, microscopy specialist
Filipa Oliveira, PhD student
Anniina Pirttiniemi, postdoc
Aino Tedeton, researcher
Yan Zhang, researcher
Yizhou Hu, researcher/bioinformatician
Schoofs H, Zhang Y, Ortsäter H, Mariya L, Benedito R, Mäkinen T. VEGFR2 is required for VEGF-C–VEGFR3–PI3Kα-mediated sprouting lymphangiogenesis. Nat Commun 2026; in press.
Kraft M, Schoofs H, Petkova M, Andrade J, Grosso AR, Benedito R, De Roo AK, Boon LM, Vikkula M, Kapp FG, Hägerling R, Potente M, Mäkinen T. Angiopoietin-TIE2 feedforward circuit promotes PIK3CA-driven venous malformations. Nat Cardiovasc Res 2025; Jul;4(7):801-820. doi: 10.1038/s44161-025-00655-9.
Schoofs H, Daubel N, Schnabellehner S, Grönloh MLB, Palacios Martínez S, Halme A, Marks AM, Jeansson M, Barcos S, Brakebusch C, Benedito R, Engelhardt B, Vestweber D, Gaengel K, Linsenmeier F, Schürmann S, Saharinen P, van Buul JD, Friedrich O, Smith RS, Majda M, Mäkinen T. Dynamic cytoskeletal regulation of cell shape supports resilience of lymphatic endothelium. Nature 2025; May;641(8062):465-475. doi: 10.1038/s41586-025-08724-6.
Petkova M, Kraft M, Stritt S, Martinez-Corral I, Ortsäter H, Vanlandewijck M, Jakic B, Baselga E, Castillo SD, Graupera M, Betsholtz C, Mäkinen T. Immune-interacting lymphatic endothelial subtype at capillary terminals drives lymphatic malformation. J Exp Med 2023; 220(4):e20220741. doi: 10.1084/jem.20220741.
Hernández Vásquez MN, Ulvmar MH, González-Loyola A, Kritikos I, Sun Y, He L, Halin C, Petrova TV, Mäkinen T. Transcription factor FOXP2 is a flow-induced regulator of collecting lymphatic vessels. EMBO J 2021; 40(12):e107192. doi: 10.15252/embj.2020107192
Frye M, Stritt S, Ortsäter H, Hernandez-Vasquez M, Kaakinen M, Vicente A, Wiseman J, Eklund L, Martinez-Torrecuadrada JL, Vestweber D, Mäkinen T. EphrinB2-EPHB4 signalling provides Rho-mediated homeostatic control of lymphatic endothelial cell junction integrity. eLife 2020; 9:e57732.
Martinez-Corral I, Zhang Y, Petkova M, Ortsäter H, Sjöberg S, Diez SC, Brouillard P, Libbrecht L, Graupera M, Alitalo K, Boon L, Vikkula M, Mäkinen T. Blockade of VEGF-C signaling inhibits lymphatic malformations driven by oncogenic PIK3CA mutation. Nat Commun 2020; 11:2869. doi: 10.1038/s41467-020-16496-y
Frye M, Taddei A, Dierkes C, Martinez-Corral I, Fielden M, Ortsäter H, Kazenwadel J, Calado DP, Ostergaard P, Salminen M, He L, Harvey N, Kiefer F, Mäkinen T. Matrix stiffness controls lymphatic vessel formation through regulation of a GATA2-dependent transcriptional program. Nat Commun 2018; 9:1511. doi: 10.1038/s41467-018-03959-6
Zhang Y, Ulvmar MH, Stanczuk L, Martinez-Corral I, Frye M, Alitalo, K, Mäkinen T. Heterogeneity in VEGFR3 levels drives lymphatic vessel hyperplasia through cell-autonomous and non-cell-autonomous mechanisms. Nat Commun 2018; 9:1296. doi: 10.1038/s41467-018-03692-0
Full list
https://www.webofscience.com/wos/author/record/C-5765-2014
Jenny and Antti Wihuri Foundation
Research Council of Finland
Sigrid Juselius Foundation
Cancer Foundation Finland