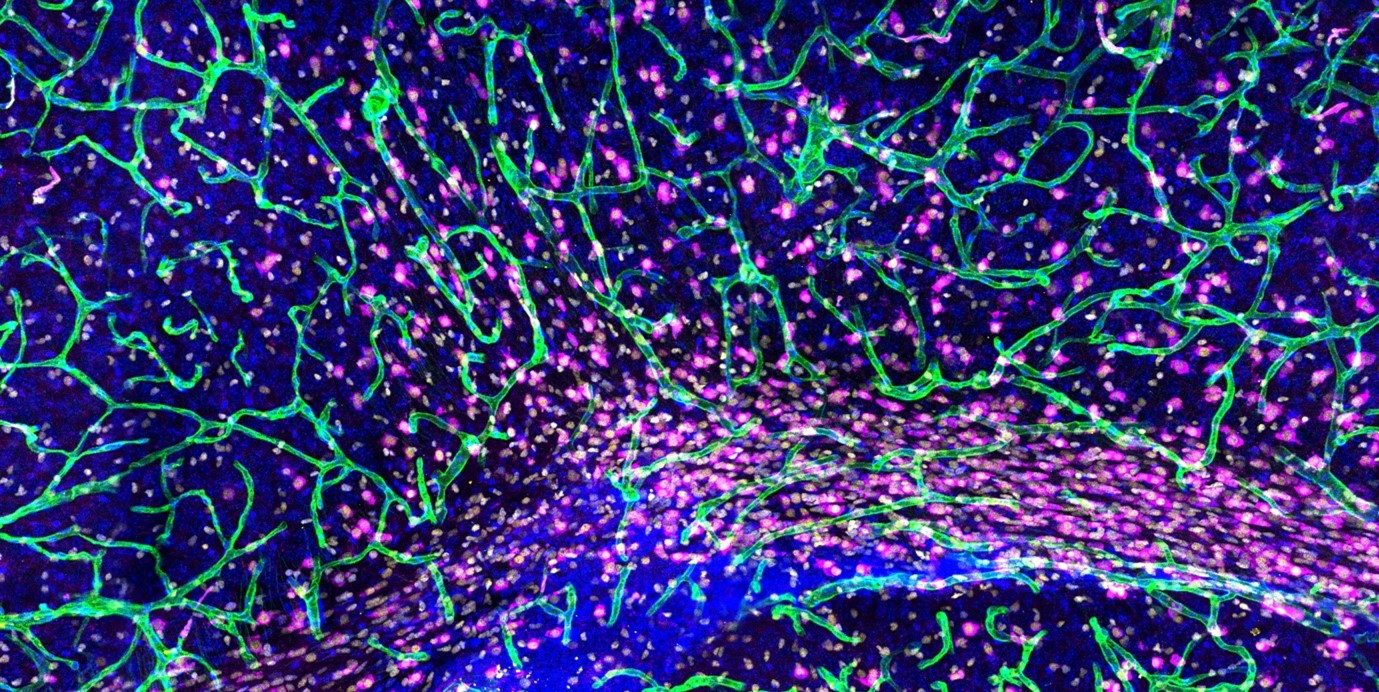
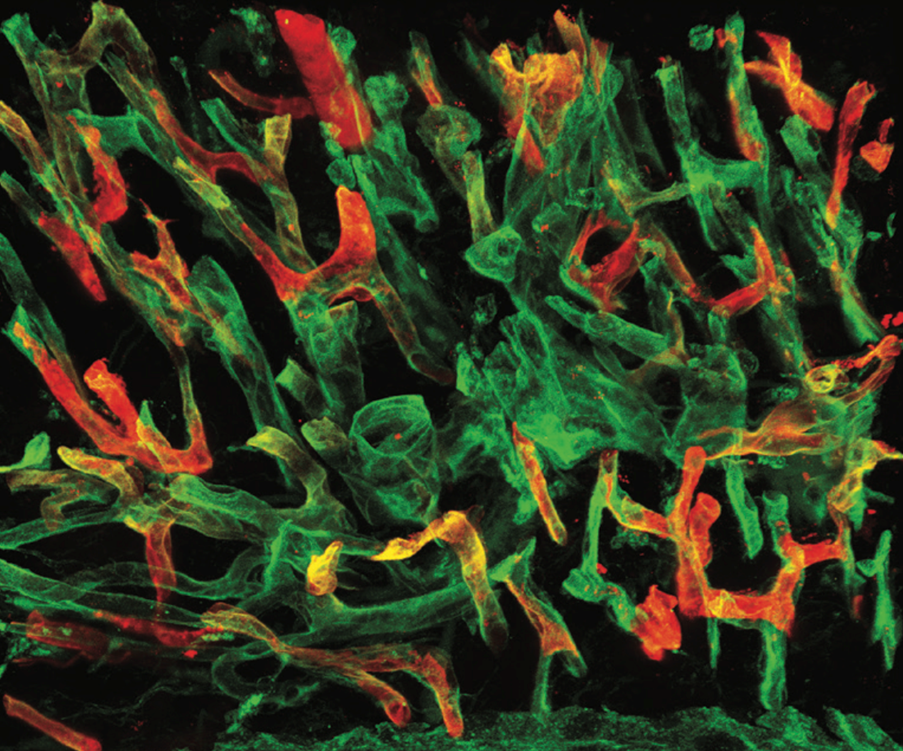
We study how blood and lymphatic vessels regulate central nervous system homeostasis and white matter integrity, focusing on vascular mechanisms that shape oligodendrocyte lineage cell function across development, aging, and repair.
Blood and lymphatic vessels, lined by endothelial cells, are essential for maintaining central nervous system (CNS) homeostasis. Endothelial cells regulate barrier properties, molecular exchange, metabolic balance, and immune responses, and release angiocrine factors that signal to surrounding cells, creating the controlled environment required for proper neural and glial function. Disturbances in these vascular systems, including those that arise with aging or inflammation, can impair tissue homeostasis, limit repair capacity, and contribute to neurological diseases. Yet the mechanisms by which blood and lymphatic vessels support and communicate with CNS resident cells remain incompletely understood.
We investigate how blood and lymphatic vessels regulate oligodendrocyte lineage cells, the progenitor and myelinating cells responsible for maintaining white matter integrity. We are particularly interested in how vascular-derived cues shape the oligovascular niche and influence oligodendrocyte precursor cell function, oligodendrocyte maturation, myelin maintenance, and repair. By studying these mechanisms across development, homeostasis, aging, and injury, we aim to understand how vascular dysfunction contributes to white matter vulnerability and to identify pathways that could support myelin maintenance and enhance white matter recovery in aging and neurological disorders.
Current Group members
Anna-Maissoun Al Khani, MSc, PhD student
Nathaniel Langan, MSc, PhD student
Korhonen EA, Murtomäki A, Jha SK, Anisimov A, Pink A, Zhang Y, Stritt S, Liaqat I, Stanczuk L, Alderfer L, Sun Z, Kapiainen E, Singh A, Sultan I, Lantta A, Leppänen VM, Eklund L, He Y, Augustin HG, Vaahtomeri K, Saharinen P, Mäkinen T, Alitalo K. Lymphangiogenesis requires Ang2/Tie/PI3K signaling for VEGFR3 cell-surface expression. J Clin Invest. 2022;132(15):e155478.
Leppänen VM*, Brouillard P*, Korhonen EA, Sipilä T, Kumar Jha S, Revencu N, Labarque V, Fastré E, Schlögel M, Ravoe M, Singer A, Luzzatto C, Angelone D, Giovanni Crichiutti G, D’Elia A, Kuurne J, Elamaa H, Koh GY, Saharinen P, Vikkula M#, Alitalo K#. Characterization of ANGPT2 mutations associated with primary lymphedema. Sci Transl Med. 2020;12:eaax8013. * # Equal contribution
Li Z*,Korhonen EA*, Merlini A, Strauss J, Wihuri E, Nurmi H, Antila S, Paech J, Deutsch U, Engelhardt B, Chintharlapalli S, Koh GY, Flügel A, Alitalo K. Angiopoietin-2 blockade ameliorates autoimmune neuroinflammation by inhibiting leukocyte recruitment into the CNS. J Clin Invest. 2020;130:1977-1990. * Equal contribution
Korhonen EA*, Lampinen A*, Giri H, Anisimov A, Kim M, Allen B, Fang S, D’Amico G, Sipilä TJ, Lohela M, Strandin T, Vaheri A, Ylä-Herttuala S, Koh GY, McDonald DM, Alitalo K#, Saharinen P#. Tie1 controls angiopoietin function in vascular remodeling and inflammation. J Clin Invest. 2016;126:3495-510. * # Equal contribution
D’Amico G, Korhonen EA, Anisimov A, Zarkada G, Holopainen T, Hägerling R, Kiefer F, Eklund L, Sormunen R, Elamaa H, Brekken RA, Adams RH, Koh GY, Saharinen P and Alitalo K. Tie1 deletion inhibits tumor growth and improves angiopoietin antagonist therapy. J Clin Invest. 2014;124:824-834.