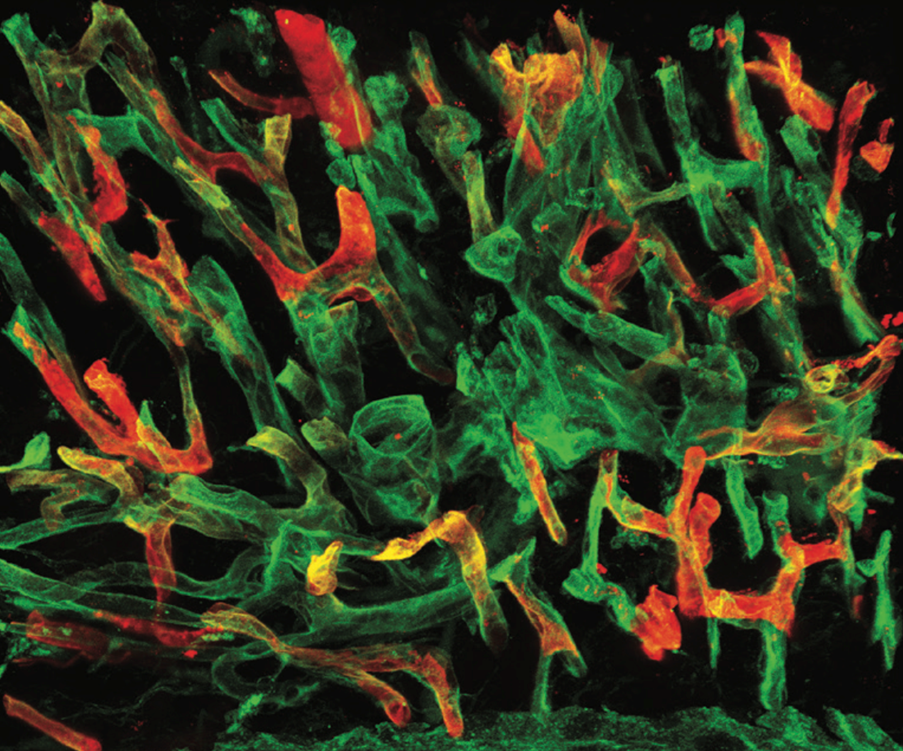
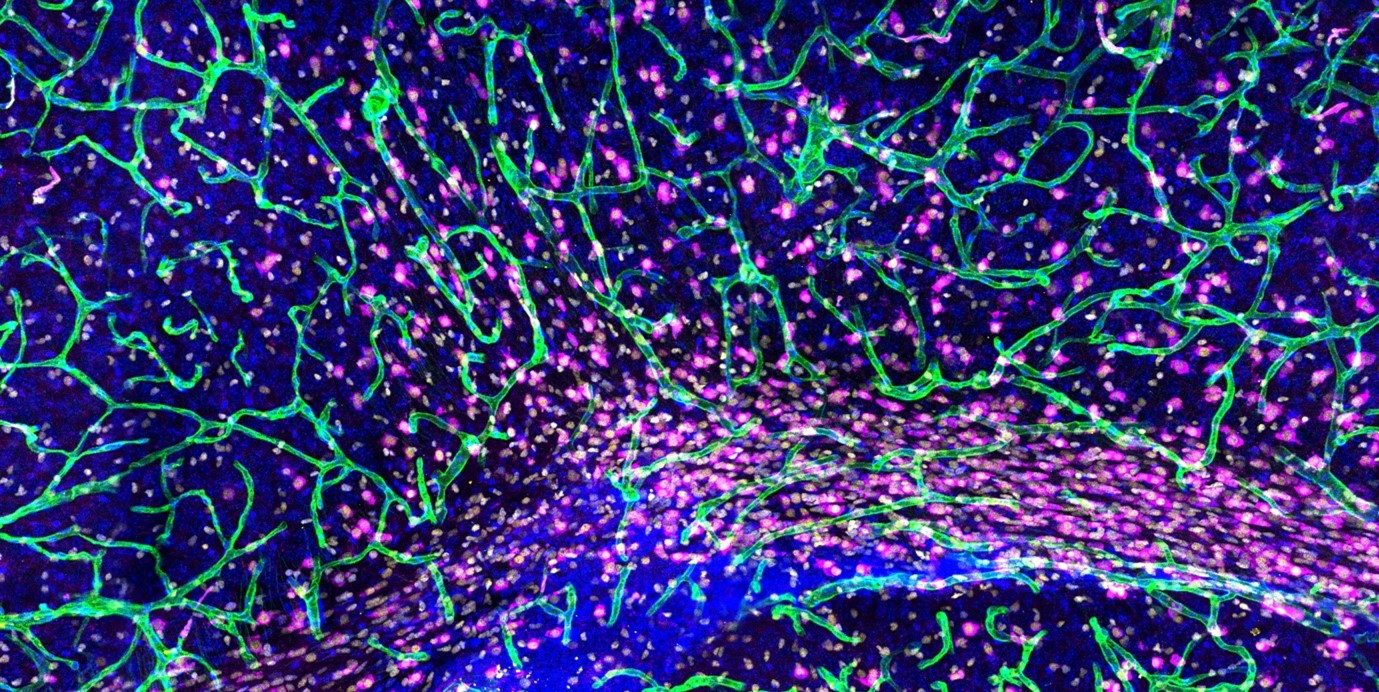
We study the fundamental mechanisms that govern the development, maintenance, and regeneration of a functional lymphatic vasculature. Additionally, we investigate the role of lymphatic vessels in organ-specific physiology and disease processes.
The lymphatic vasculature is increasingly recognized for its role as a multifaceted regulator of tissue homeostasis and regeneration. Traditionally, its primary function has been to drain fluid, macromolecules, and immune cells from peripheral tissues into the systemic circulation via lymph nodes. Dysfunction of lymphatic vessels can lead to the accumulation of protein-rich fluid in tissues, known as lymphedema, and impaired immune responses.
Recent findings have revealed additional roles of the lymphatic system, including active modulation of adaptive immunity by lymphatic endothelial cells (LECs) and the secretion of paracrine (lymphangiocrine) factors that regulate organ growth and regeneration. The expanding understanding of the diverse functions of the lymphatic system in essential physiological processes and disease conditions, such as autoimmune disease and atherosclerosis, underscores the need for a better understanding of the underlying mechanisms.
Our research seeks to comprehensively explore the fundamental mechanisms that regulate the lymphatic vasculature and its role in organ-specific physiology and disease processes. This knowledge is critical for understanding pathological alterations in lymphatic vessels that contribute to the onset and progression of diseases, providing opportunities for the development of novel therapies.
Current Group members
Gauri Arolkar, PhD student
Vishal Mohanakrishnan, postdoc
Yan Zhang, researcher
Yizhou Hu, researcher/bioinformatician
Uppsala
Bojana Jakic, postdoc
Charlotte Rorsman, senior technician
Filipa Oliveira, PhD student
Ingvar Ferby, researcher
Sarah Schnabellehner, postdoc
Sofie Lunell Segerqvist, laboratory assistant
Zuzana Varaliova, postdoc
Schoofs H, Daubel N, Schnabellehner S, Grönloh MLB, Palacios Martínez S, Halme A, Marks AM, Jeansson M, Barcos S, Brakebusch C, Benedito R, Engelhardt B, Vestweber D, Gängel K, Linsenmeier F, Schürmann S, Saharinen P, van Buul JD, Friedrich O, Smith RS, Majda M, Mäkinen T. Dynamic cytoskeletal regulation of cell shape supports resilience of lymphatic endothelium. Nature in press (2025).
Petkova M, Kraft M, Stritt S, Martinez-Corral I, Ortsäter H, Vanlandewijck M, Jakic B, Baselga E, Castillo SD, Graupera M, Betsholtz C, Mäkinen T. Immune-interacting lymphatic endothelial subtype at capillary terminals drives lymphatic malformation. J Exp Med Apr 3;220(4):e20220741. doi: 10.1084/jem.20220741 (2023).
Hernández Vásquez MN, Ulvmar MH, González-Loyola A, Kritikos I, Sun Y, He L, Halin C, Petrova TV, Mäkinen T. Transcription factor FOXP2 is a flow-induced regulator of collecting lymphatic vessels. EMBO J 40(12):e107192. doi: 10.15252/embj.2020107192 (2021).
Frye M, Stritt S, Ortsäter H, Hernandez-Vasquez M, Kaakinen M, Vicente A, Wiseman J, Eklund L, Martinez-Torrecuadrada JL, Vestweber D, Mäkinen T. EphrinB2-EPHB4 signalling provides Rho-mediated homeostatic control of lymphatic endothelial cell junction integrity. eLife 9:e57732 (2020).
Martinez-Corral I, Zhang Y, Petkova M, Ortsäter H, Sjöberg S, Diez SC, Brouillard P, Libbrecht L, Graupera M, Alitalo K, Boon L, Vikkula M, Mäkinen T. Blockade of VEGF-C signaling inhibits lymphatic malformations driven by oncogenic PIK3CA mutation. Nat Commun 11:2869 doi: 10.1038/s41467-020-16496-y (2020).
Frye M, Taddei A, Dierkes C, Martinez-Corral I, Fielden M, Ortsäter H, Kazenwadel J, Calado DP, Ostergaard P, Salminen M, He L, Harvey N, Kiefer F, Mäkinen T. Matrix stiffness controls lymphatic vessel formation through regulation of a GATA2-dependent transcriptional program. Nat Commun, 9:1511 doi: 10.1038/s41467-018-03959-6 (2018).
Zhang Y, Ulvmar MH, Stanczuk L, Martinez-Corral I, Frye M, Alitalo, K, Mäkinen T. Heterogeneity in VEGFR3 levels drives lymphatic vessel hyperplasia through cell-autonomous and non-cell-autonomous mechanisms. Nat Commun 9:1296 doi: 10.1038/s41467-018-03692-0 (2018).
Martinez-Corral I, Ulvmar MH, Stanczuk L, Tatin F, Kizhatil K, John SWM, Alitalo K, Ortega S, Makinen T. Non-venous origin of dermal lymphatic vasculature. Circ Res 116:1649-1654 (2015).
Stanczuk L, Martinez-Corral I, Ulvmar MH, Zhang Y, Lavina B, Fruttiger M, Adams RH, Saur D, Betsholtz C, Ortega S, Alitalo K, Graupera M, Mäkinen T. cKit lineage hemogenic endothelium-derived cells contribute to mesenteric lymphatic vessels. Cell Rep 10:1708-1721 (2015).
Tatin F, Taddei A, Weston A, Fuchs E, Devenport D, Tissir F, Makinen T. Planar cell polarity protein Celsr1 regulates endothelial adherens junctions and directed cell rearrangements during lymphatic valve morphogenesis. Dev Cell 15:31-44 (2013).
Lutter S, Xie S, Tatin F, Makinen T. Smooth muscle-endothelial cell communication activates Reelin signaling and regulates lymphatic vessel formation. J Cell Biol 197:837-49 (2012).
Bazigou E, Lyons OTA, Smith A, Venn GE, Cope C, Brown NA, Makinen T. Genes regulating lymphangiogenesis control venous valve formation and maintenance in mice. J Clin Invest 121:2984-92 (2011).
Bazigou E, Xie S, Chen C, Weston A, Miura N, Sorokin L, Adams R, Muro A, Sheppard D, Makinen T. Integrin-alpha9 is required for fibronectin matrix assembly during lymphatic valve morphogenesis. Dev Cell 17:175-186 (2009).
Full list
https://www.webofscience.com/wos/author/record/C-5765-2014
No funding information